How We Can Help You
Te Ira Kāwai, the Auckland Regional Tissue Bank (ARTB), collects and stores human tissues for use by researchers in ethically approved research.
Te Ira Kāwai (ARTB) aims to assist researchers with their research projects and build a bank of valuable tissue (including blood) at the same time.
ARTB is a University of Auckland / metro Auckland DHB research platform and all researchers are strongly advised to engage and involve the ARTB in all projects using human tissue
ARTB uses OpenSpecimen, a specialist Biobank database, which enables us to track storage locations and individual samples efficiently to ensure banked tissues can be monitored at all times and to minimize the risk of sample loss.
We have a large collection of banked human tissue that has been collected for Future Unspecified Research (FUR).
A Researcher can request to use tissues from the tissue bank for their specific study under its own ethical approval. This is often referred to as “Pathway 1”
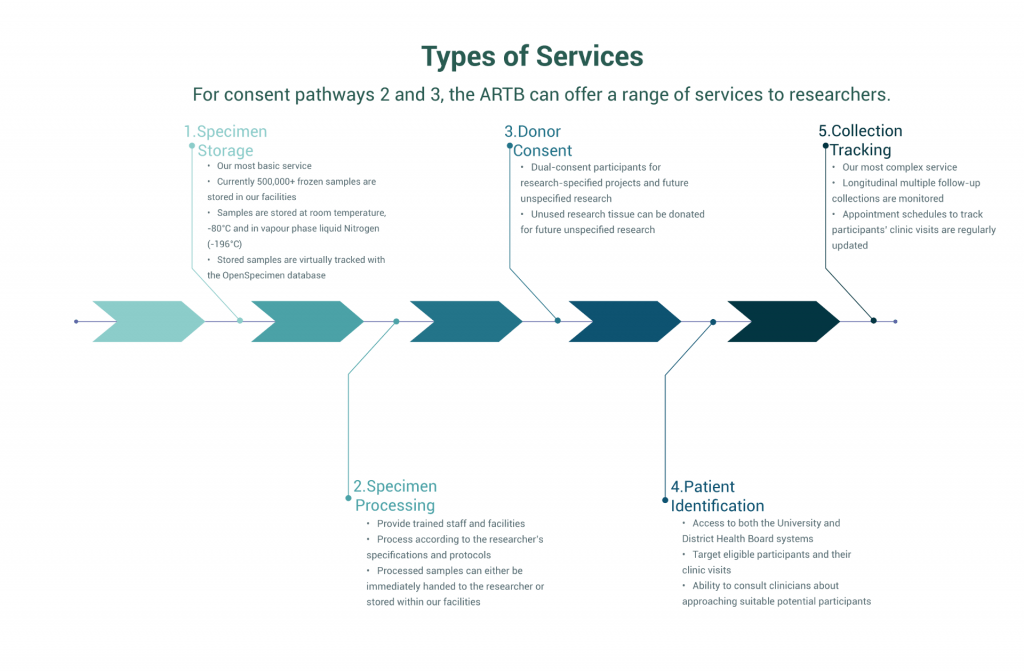
Alternatively the Tissue Bank can work with Researchers to collect, process and store tissue specifically for their research projects
This will ensure that samples are collected in a ‘bankable’ manner using the correct database tracking and storage tubes from the outset. There are two pathways that can be followed to collect tissue for specific projects
We help researchers collect tissues for their specific ethically approved research studies, while at the same time banking some of the donated tissue for FUR often referred to as “Pathway 2”
This is the most cost effective and least complicated way to collect high quality tissues for a specific study, while also storing tissue for future ethically approved studies. (FUR)
The tissue banked for FUR will then form part of the ARTB and is available for any research project in the future as long as it is ethically approved and has met the access criteria
We can collect, process and store tissue for specific research projects when FUR is not feasible. This is known as "Pathway 3"
This is a service to University and DHB researchers. ARTB works with your research team to facilitate the collection and storage of tissue for a specific research project. Tissue samples are not retained for FUR. The project must be governed by an ethics approval that has been granted by an approved ethics committee to a named researcher.
While we need to charge a fee for this service, researchers have found this fee is less than the cost of employing their own staff to identify donors and collect tissues or blood along with clinical information.
Guide to developing a budget when the ARTB doesn’t already hold a collection of samples you need for your project
ARB Services Price List
| Core Services | ||
| Patient consent | Per hour | $94.08 |
| Sample collection (technician) | Per hour | $94.08 |
| Sample collection (research nurse) | Per hour | $105.83 |
| Sample processing | Per hour | $94.08 |
| Sample Storage | ||
| -80 storage (per rack per year) | $11.76 | |
| -196 storage (per rack per year) | $17.64 | |
| OpenSpecimen sample & data management | Per hour | $94.08 |
| Collection protocol setup | Per hour | $94.08 |
| Support and training to use OpenSpecimen | One-off fee | $1528.65 |
| Additional Services | ||
| Microtomy | Per block | $- |
| Tissue Mark Up | Per H&E slide | $- |
| H&E Slide | Per slide | $- |
| Histology service charge (in addition of the above) | Per hour | $- |
| Clinical data provision | Per hour | $94.08 |
| Administrative cost | Per application | $235.18 |
Information regarding Ethics Approvals for projects using human tissue
If your research involves collecting tissue directly from a donor rather than applying to access tissue that has already been banked, it requires a specific study related ethical approval
The Health and Disability Ethics Committees (HDEC) have recently moved towards requesting all tissues “left over” from a specific study that can be retained for future unspecified research (FUR), must be moved into a HDEC-approved tissue bank, such as the ARTB, at the end of each research study.
If you discuss your study with the ARTB before you submit your ethics application we can help with both your study specific ethics application and also if necessary how to build in “dual consent” so a part of the collected tissue is stored for Future Unspecified Research (FUR)
Ethical approvals can currently be obtained through several routes, therefore we recommend consulting an ethics adviser in your organisation before planning your research study.
Some research requires approval from our national Health and Disability Ethics Committees (HDEC).
HDEC review will usually be required if:
- samples are not de-identified for research
- studies are at higher risk where patients are to be identified or, a large clinical datasets are required
- there is a possibility of generating genetic incidental findings that may be of relevance to the donor or their families.
Studies that do not require HDEC review but do involve staff and/or patients from the Auckland and Counties Manukau District Health Boards will need to be submitted to The Auckland Health Research Ethics Committee (AHREC).
For University of Auckland staff, details of the application process can be found at https://www.auckland.ac.nz/en/about/research/re-ethics/auckland-health-research-committee.html#1d6188eeb079e69f61d15f5bbe279ffe .